Who we are
One of Australia’s few TGA-licensed, Phase 3–ready GMP facilities dedicated to cell therapy manufacturing
Deep expertise in MSC manufacturing, underpinned by robust GMP infrastructure and integrated QC laboratories
Broader capability spanning iPSCs, NK cells, CAR-T, exosomes, umbilical cord–derived cells, and fibroblasts
End-to-end support from preclinical development through to pivotal clinical supply
Right-sized CDMO focus — prioritised attention, fair pricing, and responsive timelines for emerging innovators
Purpose-built facility with scalable cleanrooms and adjacent warehouse capacity for future GMP expansion
Our history
Preclinical since 2009
GLP established for early Phase 2014
Site established Jan 2022
Construction start Mid 2022
Construction finished Feb 2023
Inspection Dec 2023
License granted April 2025
Core services
01
Biobanking
- MCB & WCB generation
- Cell characterisation
02
Process development
- Process optimisation for GMP
- Aseptic process validation
- Scale up process development
03
GMP manufacturing
- Tech transfer framework
- Aseptic protocol development and validation
- Engineering runs
- GMP manufacturing (small to large scale)
- Downstream processing
04
Quality assurance and control MCB & WCB generation
- Validation protocol and SOP development
- TGA audit support if required
- QC method validation
05
Regulatory dossiere support
- CMC document support and advice
- Access to regulatory support if required
06
Storage and logistics
- Validated control rate freezing
- Liquid nitrogen freezer
Domestic & International shipping. Room temp to coldchain
Our facility is more than just a GMP space – it’s a platform for your success. With flexible, scalable manufacturing suites, integrated QC labs and Phase 3 approval, we provide a trusted environment where cell therapies are developed with precision, speed, and uncompromising quality.
Clean rooms available
- State of the art clean rooms Grade A, Grade B, Grade C, Grade D available to suit your needs.
Qualified equipment available to suit your needs
- Multigas incubators
- Water bath
- Centrifuge (refrigerated)
- Bioreactor
- BSC’s
- Particle counters
- Control rate freezer
- Fridges
- Freezers
- Biowelder
- Full list available upon request.
- Back up generator to ensure continuity of service
Integrated Quality Control laboratories – in-house sterility, identity, and potency testing.
- Scalability
With adjacent warehouse capacity, our facility is designed to scale rapidly, enabling expansion into new GMP manufacturing space without disruption.
Quality test menu
| Test Item | Details |
|---|---|
| ELISA | Biomolecule quantification assay used to detect proteins, antibodies and hormones |
| FACS (flow cytometry) | Uses light scattering and fluorescence to sort and characterise cell populations for expressed markers, size, phenotype and cell life cycle analysis |
| Endotoxin | Detects bacterial endotoxins that may cause adverse immune reactions |
| Mycoplasma | Sensitive assay for detecting hidden mycoplasma contamination |
| Cell differentiation capability | Confirms cells identity & multipotency though differentiation assays |
| Cell count & viability | Ensure accurate dosing and cell health prior to release |
| pH | Tests product acidity or basic properties |
| EM (environmental monitoring) | Continuous assessment of cleanroom air and surfaces to ensure GMP compliance |
| Gram stain | Rapid screen for bacterial or fungal contamination |
| Growth promotion tests | Confirms culture media/EM plates can support microbial growth |
| Bioburden | Detect and quantifies microbial contamination in cell products and solutions |
| QPCR/ddPCR | Sensitive genetic assay for detecting viral or microbial contaminant |
A phased approach to manufacturing and scalability
Pre-clinical
Hands on experience with MSC’s, IPSC’s, NK cells
Phase 1
Early GMP manufacturing to support first–in–human studies. We provide tech transfer, access to partnered clinical sites, small–scale GLP/GMP production, and tailored QC assays, ensuring your therapy enters the clinic with a robust and compliant foundation
Phase 2
In Phase 2, we focus on scaling processes and providing access to clinical partners to ensure compliance for larger patient cohorts. Our team delivers GMP–compliant manufacturing, validated QC assays, and robust tech transfer plans, providing a smooth transition into our facility and laying the groundwork for late-stage development and GMP manufacturing compliance
Phase 3
At Phase 3, we provide Phase 3–licensed GMP manufacturing with the capacity and rigor required for pivotal trials. Our expertise in scale-up, regulatory CMC documentation, TGA audited QMS, reliability and readiness for commercial supply ensures consistency
Commercial
We provide bioreactor 3D-cell-culture technology process co-development (18 months) Manufacturing line and facility/room build up (18 months) ER, APV, Stability (12–18 months). Inspection and audit ready
Manufacturing capability
Pre-clinical and Phase 1
T-flask/shaker flasks/high throughout screening with DoE
2 & 4 level cell factory
Centrifuge
Manual filling
Manufacturing capability
Phase 2 onwards
Small scale bioreactor 3.75L
Medium scale bioreactor
Large scale bioreactor 40L
Automated fill and finish
A structure dedicated to your account
Magellan is Australia-based so your project may be eligible for the 43.5% Commonwealth Government R&D tax rebate. We would be happy to discuss this with you as part of project planning.
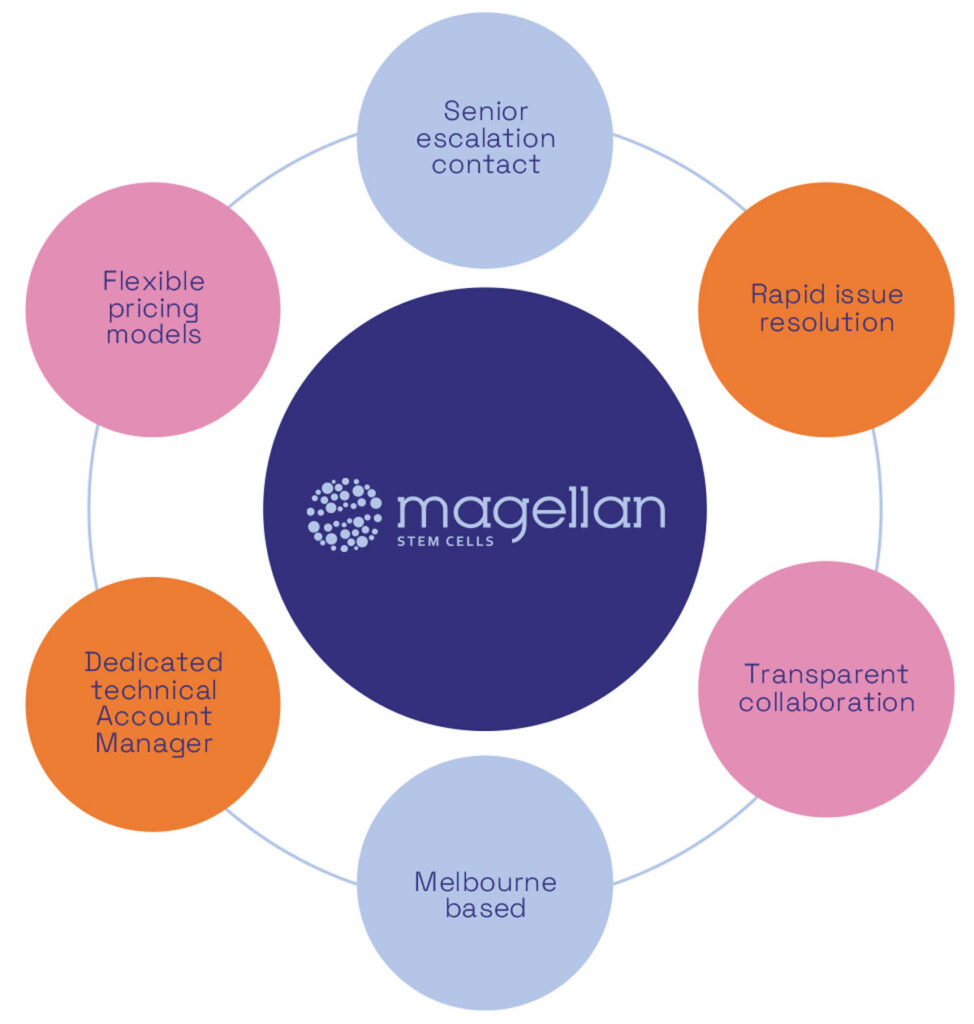
Why partner with us
- End-to-end cell therapy support from concept through Phase 3 GMP manufacturing
- Phase 3-ready, TGA-licensed infrastructure designed for late-stage clinical supply
- Right-sized CDMO — large enough to deliver, small enough to prioritise your program
- Fair and transparent pricing models for university and emerging small to mid-stage innovators
- Flexible, scalable processes for adherent and non-adherent platforms T-flasks, cell factories, 3.75L bioreactors, 10L bioreactors, 40L bioreactors, fill and finish
- Dedicated technical account management with direct access to decision-makers to ensure seamless execution of your project
- Agile scheduling and rapid responsiveness — your therapy never waits in a queue

Nick Bliesner
Chief Operating Officer